Recall: Teething Rings Recalled by Whitehall-Robins Healthcare
| FOR IMMEDIATE RELEASE June 29, 2000 Release # 00-135 |
Teether Recall Hotline: (800)525-2607 CPSC Consumer Hotline: (800) 638-2772 CPSC Media Contact: Nychelle Fleming, (301) 504-7063 |
CPSC, Whitehall-Robins Healthcare Announce Recall of Teething Rings
WASHINGTON, D.C. - In cooperation with the U.S. Consumer Product Safety Commission (CPSC), Whitehall-Robins Healthcare, of Madison, N.J., is voluntarily recalling about 475,000 teething rings. When bent, these teething rings can fit into an infant's mouth and trigger a gagging reflex. This poses a risk of vomiting, which could result in choking and aspiration.Whitehall-Robins Healthcare has received two reports from consumers of children gagging on the teething rings, including one report of a child starting to choke.
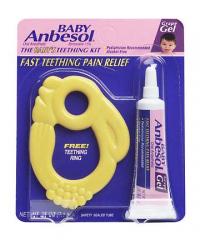
These teethers were sold with .25 oz. tubes of Baby Anbesol® Grape Gel, an oral anesthetic. The teethers are yellow and shaped like rabbits. They are about 3 inches long and 2.25 inches high. Two holes are molded into the teething ring for infants to grasp. The packaging reads, "THE BABY'S TEETHING KIT," and "FAST TEETHING PAIN RELIEF."
Mass merchandise, drug and grocery stores nationwide sold Baby Anbesol with these teethers from May 1999 through June 2000 for about $6.
Consumers should take these teething rings away from infants immediately. Contact Whitehall-Robins Healthcare for information on returning the teething ring in order to receive a free Anbesol® product. Consumers can call Whitehall-Robins Healthcare at (800) 525-2607 between 9 a.m. and 5 p.m. ET Monday through Friday, or go to the firm's web site at http://www3.young-america.com/2506-2/Recall.asp.
Only the teething rings, and not the Baby Anbesol® Grape Gel, are being recalled.